Amino Acids In Agriculture
Although the majority of nitrogen fertilizers used in agriculture today are chemically synthesized to create nutrients like nitrates and ammonium, there’s growing interest in another type of nitrogen fertilizer: amino acids. These organic molecules are environmentally friendly, require far less energy to produce, can be entirely plant-derived, promote beneficial microbial activity and soil fertility, and are the most energy efficient form of nitrogen fertilizer for plants to use–ultimately leading to bigger, higher quality yields when properly implemented. But to understand how all of this is possible, it’s important to understand what amino acids are and how they can work as fertilizers.
Reduce your synthetic fertilizer use, improve plant growth, and promote soil health with organic, plant-derived amino acid-containing formulations like Lumina™. Click here to learn more!
What are Amino Acids?
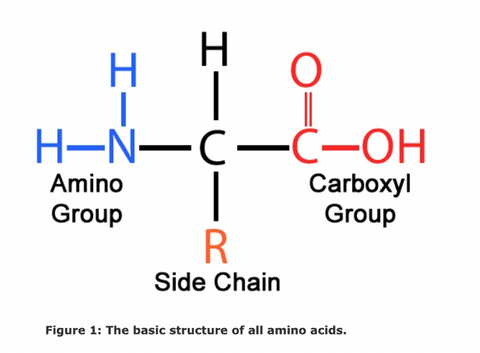
Amino acids are organic compounds that make up proteins in all living things. All amino acids are made up of a central carbon, an amino group containing nitrogen, and a variable side chain which determines the different amino acid characteristics, like pH, polarity, and charge. In addition to forming proteins, amino acids also play a role in regulating different metabolic processes and serve as precursors for plant hormones.
(Image credit: NASA AMES Research Center)
How do Amino Acids Compare to Synthetic Nitrogen Fertilizers?
Synthetic fertilizers like calcium nitrate, ammonium phosphate, urea, and ammonium nitrate all rely on the energy-intensive Haber-Bosch process to create ammonia (NH3) from atmospheric nitrogen and metal catalysts. Unfortunately, this process is hugely energy intensive, consuming approximately 1-2% of the world’s energy supply each year and contributing about 1% of total CO2 emissions globally just in its manufacturing. Worse yet, these synthetic fertilizers contribute massively to soil and water pollution, but are still frequently used due to their low cost and high nitrogen content.
Ultimately, the synthetic fertilizers produced from the Haber-Bosch process, like nitrates (NO3) and ammonia (NH3), are simple molecules that require biochemical reactions from plants in order to be effectively utilized. In fact, these molecules are ultimately used by the plant to create amino acids! For that reason, applying amino acids directly as a fertilizer source can save the plant an energetic step and be immediately absorbed and utilized to promote growth. Amino acids are also excellent chelating agents, meaning they can help the plant uptake micronutrients in addition to providing a great source of nitrogen.
Amino Acids in Plants
Like all living organisms, plants need essential amino acids to make proteins, which make up intra- and extracellular structures, regulate metabolic processes, and facilitate nutrient transport. Amino acids can be taken up through the roots or directly through the stoma on the leaves. Some examples of amino acids that are found in high quality, biostimulating fertilizers include:
Glycine, which is the simplest amino acid and only has a hydrogen atom as its side chain. Due to its simple structure, glycine can easily penetrate plant tissue and is a good source of plant-available nitrogen (Amino Acids, 2007).
Tryptophan, which is a precursor to Indole Acetic Acid (IAA), a plant growth hormone that stimulates lateral root growth (Moe, 2013). IAA and IBA are the natural analogue to the synthetic plant hormone 1-Naphthaleneacetic acid (NAA), which is commonly found in rooting hormones.
Glutamate, which is a precursor to the synthesis of several other amino acids and is involved in different metabolic pathways, so the addition of glutamate has the potential to increase the abundance of several amino acids (Teixeira et al., 2018).
Proline, which has been shown to enhance stress tolerance and is correlated with increased plant growth and yield (Ashraf and Foolad, 2007).
Why Apply Amino Acids to Crops?
Because all amino acids have a conserved structure including carbon and nitrogen, applying exogenous amino acids functions as a nitrogen and carbon source for the plant. Nitrogen is essential to plants. It is vital in chlorophyll production, a component in nucleic acids which make up DNA and RNA, and functions in energy transfer by ATP. Plants uptake nitrogen as nitrate and ammonium and through nitrogen fixation by soil microbes, however, it is more energetically efficient for the plant to take up organic nitrogen in the form of amino acids. Plants can redirect energy that would otherwise be needed to assimilate absorbed nitrogen into amino acids by using amino acids directly. This increases nitrogen metabolism and can reduce stress related to nitrate and ammonium availability, allowing for better root development. Thus, enhancing rhizosphere health and the breakdown of amino acids, generally increasing the uptake of urea and other nutrients by plant roots.
A plant’s capacity to absorb amino acids and other nutrients through its roots is correlated to the health and activity of the rhizosphere. (Teixeira et al., 2018). Glutamate receptors bind a variety of amino acids which regulate and mediate changes in root architecture, mediate stress signaling, signal carbon metabolism, stomatal movements, photosynthesis and plant immunity. Activated glutamate receptors lead to increased root development and in turn provide greater ability for nitrogen uptake and uptake of other nutrients. Overall, reducing plant stress allows for an increase in nitrate reductase activity, resulting in improved plant growth and yield (Teixeira et al., 2018).
Chelating effects are another reason to apply amino acids as a fertilizer source to crops. Metal cations found in most soils can be unavailable, and sometimes even harmful, to plants. Amino acids in the soil can bind to these metal cations, forming molecular ‘complexes’. When a cation binds to an organic compound at two or more places, it forms a chelate. This is beneficial because chelated and complexed nutrients are more available to the plant for uptake. Lysine, glutamic acid, cysteine, and histidine are some of the amino acids known to complex or chelate micronutrients in the soil, thus increasing overall nutrient availability and acquisition for plants. Calcium, copper, iron, magnesium, manganese, potassium and zinc and commonly chelated with amino acids (Amino Acids Review, 2007).
Take your crop nutrition program to the next level with essential amino acids for plants. Order Impello’s Lumina™ now to naturally promote plant growth in any growing system.
Takeaway
Amino acids may sound unfamiliar and highly technical, but in reality, their use as a fertilizer couldn’t be any easier. High quality organic and hybrid fertilizers like Biofuel™ SP and Lumina™from Impello® are abundant in amino acids and are highly soluble in water, have virtually no toxicity and phytotoxicity concerns, can be tank-mixed with other organic and conventional inputs, and promote both plant growth and soil microbial activity. Incorporating amino acids as part of a complete crop nutrition program is an excellent way to reduce reliance on synthetic fertilizers without jeopardizing crop yields, and they can promote plant growth in ways traditional fertilizers cannot.
If you have any questions about amino acids and how they’re making agriculture better, please contact us! We’re excited to talk to you. For technical questions, email tech@impellobio.com. For general questions or to place an order, email info@impellobio.com.
References
1. Teixeira WF, Fagan EB, Soares LH, Soares JN, Reichardt K and Neto DD (2018). Seed and Foliar Application of Amino Acids Improve Variables of Nitrogen Metabolism and Productivity in Soybean Crop. Front. Plant Sci. 9:396. doi: 10.3389/fpls.2018.00396
2. Amino Acids Review. (2007) https://www.ams.usda.gov/sites/default/files/media/Cysteine%20TR.pdf
3. Moe, L. (2013). AMINO ACIDS IN THE RHIZOSPHERE: FROM PLANTS TO MICROBES. American Journal of Botany 100(9): 1692–1705.
4. Ashraf, M.R. Foolad, Roles of glycine betaine and proline in improving plant abiotic stress resistance, Environmental and Experimental Botany,Volume 59, Issue 2, 2007, Pages 206-216, ISSN 0098-8472, https://doi.org/10.1016/j.envexpbot.2005.12.006. https://www.sciencedirect.com/science/article/pii/S0098847206000268
Impello Fact Checking Standards
Impello is committed to delivering content that adheres to the highest editorial standards for accuracy, sourcing, and objective analysis. We adhere to the following standards in reviewing our blog articles:
- We have a zero-tolerance policy regarding any level of plagiarism or malicious intent from our writers and contributors.
- All referenced studies and research papers must be from reputable and relevant publications, organizations or government agencies.
- All studies, quotes, and statistics used in a blog article must link to or reference the original source. The article must also clearly indicate why any statistics presented are relevant.
- We confirm the accuracy of all original insights, whether our opinion, a source’s comment, or a third-party source so as not to perpetuate myth or false statements.